ISO 13485 QMS System Built on Confluence for Medical / IVD Devices
ReadySet is a ready-to-use ISO 13485 QMS system designed for medical and IVD device companies.
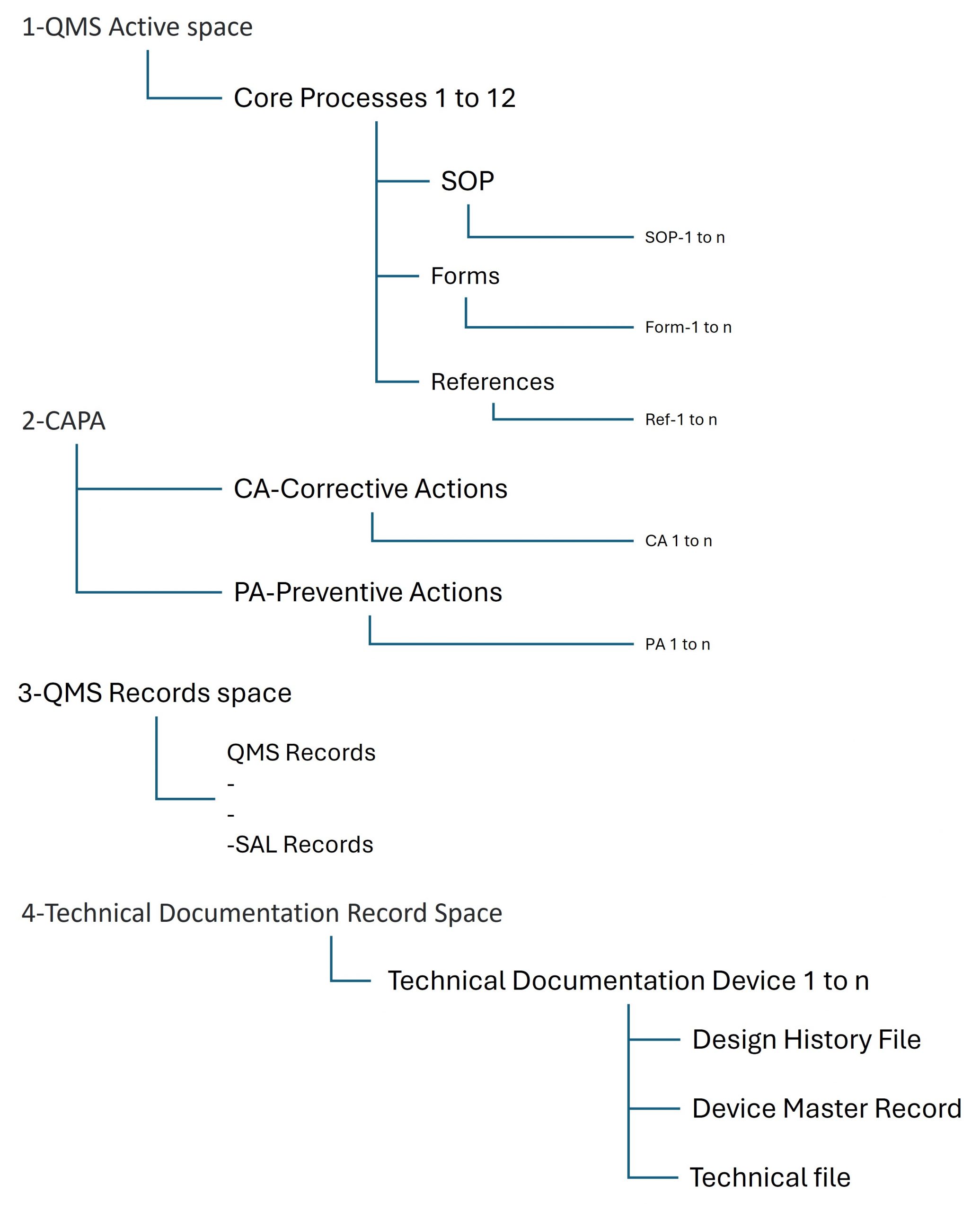
ReadySet provides a structured Quality Management System organized into dedicated spaces within Confluence.
This architecture ensures clear separation between processes, records, and technical documentation, while maintaining full traceability across the system.
The framework enables organizations to quickly deploy a complete QMS and adapt it to their product and regulatory scope.
ISO 13485 QMS on Atlassian Confluence
ReadySet is fully implemented on Atlassian Confluence, allowing teams to manage quality processes, documentation, and traceability in a collaborative environment.
QMS Active Space
The QMS Active Space contains the operational structure of the Quality Management System.
It includes the core processes required to operate an ISO 13485-compliant quality system, organized into the 12 core QMS processes shown in your current product material.
Each process includes three main types of controlled documentation:
• Standard Operating Procedures (SOPs) describing the process
• Forms used to capture data and generate quality records
• References, including internal and regulatory reference documents
The risk analysis is also managed within the QMS Active Space through dedicated forms.
This allows risk-related activities to be embedded directly into the operational QMS, with structured inputs, assessments and traceable outputs linked to the relevant processes.
This approach keeps the system practical, auditable, and easy to maintain.
CAPA and Continuous Improvement
ReadySet includes a dedicated CAPA space for managing corrective and preventive actions.
This structure supports the full lifecycle of CAPA activities:
• issue identification
• investigation and root cause analysis
• corrective actions
• preventive actions
• follow-up and effectiveness checks
This ensures a traceable and auditable improvement process.
QMS Records
The QMS Records space stores the records generated through the execution of QMS processes and forms.
These records provide objective evidence that procedures have been implemented and followed.
Typical records may include:
• training records
• supplier records
• management review records
• operational and quality records
This dedicated space helps maintain clear separation between active procedures and retained evidence.
Technical Documentation
ReadySet also includes a dedicated space for managing technical documentation for one or more devices.
For each device, the structure can include:
• Design History File (DHF)
• Device Master Record (DMR)
• Technical File
This enables separating product documentation from the core QMS while preserving consistency and traceability across the entire system.
Illustration of the ReadySet QMS architecture
This detailed video explains the ReadySet architecture and shows how to deploy and use the framework in Confluence.
Why this structure works
This architecture provides several important benefits:
• clear separation between procedures, records and technical documentation
• integrated risk analysis within the operational QMS
• easier navigation and document management
• better traceability across processes and forms
• faster preparation for audits and certification
• scalability as the company grows
The result is a practical and operational Quality Management System, not just a collection of documents.
